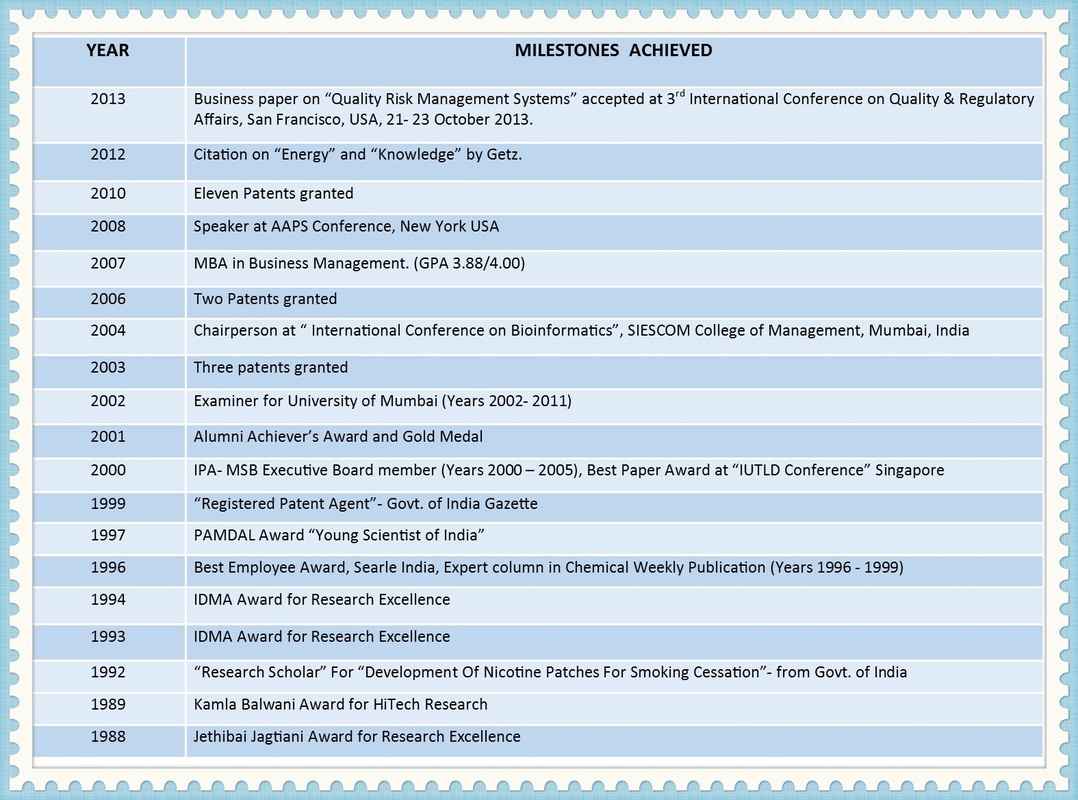
Dr. Shruti U Bhat
New Product Development and Continuous Improvement Director.


http://www.drshrutibhat.com/
http://www.drshrutibhat.com/blog
ca.linkedin.com/in/drshrutibhat
http://pharmaceuticalcareerdevelopment.blogspot.in/2010/05/welcome-to-dr-shruti-bhats.html
Industry verticals- Pharmaceuticals, Biotech, Devices, OTC, Cosmetics, Natural Health Products, Education, Management Consulting firms.
 CALGARY
CALGARYABOUT HER
Shruti U. Bhat, PHD, MBA is Innovation and Continuous Improvement Director.
She leads path-breaking product development programs such as oncology, biosimilars, nano technology and platform delivery systems for pharmaceuticals and natural products.
Shruti helps translate brand vision into product development strategies. Her mantra is to "Shorten development timelines, build quality-by-design, lean processes and bring products fast- to- market".
She works with start ups, mid-sized and growing firms in Canada, USA, Gulf and Emerging markets. She has numerous patents and publications to her credit and is an invited speaker at several international conferences and workshops.

Current

Director Product Development & Continuous Improvement.

Innoworks Inc. From oct 2012
PDFVice President Strategic Planning and Innovative Research
Getz
Director R&D and Clinical Manufacturing
PMS, Canada
Group Head Pharmaceutical R&D
Novartis
General Manager Pharma Technology Development
Unichem
Head R&D and Patents
Macleods Pharmaceuticals Limited
Asst. Manager R&D
RPG Life Sciences Limited.
Executive Applications
Klenzaids Engineers (P) Ltd.

CALGARY
.jpg)
EDUCATION HISTORY
Southern New Hampshire University (SNHU) USA-ITM
MBA, - Business Administration (GPA 3.88)
.Dale Carnegie Training Institute’s ‘Communication Skills for Managers’Patent Agent
K.M.Kundnani College of Pharmacy
M.Pharm., Ph.D.

Dr. Shruti U. Bhat, is Innovation, New Product Development and Continuous Improvement Expert.
She has a PhD in Pharmaceutical Technology and MBA. She is a gold medalist and recipient of numerous awards and certificates of merit, including Pharmaceutical industry's coveted "Young Scientist Award". She is also an invited speaker at several international conferences and workshops and writes 'Expert' columns for industry magazines, journals and newspapers worldwide. 
VANCOUVER

AREAS OF EXPERTISE
|
Video CV of Dr. Shruti U Bhat
|
Leadership And Change Management
One of her many illustrious achievements, is successful development of a Start-up R&D unit into reputable international research base, driving sales to over $ 5M within 3 years. She has also designed several award winning, quality compliant, cost- effective R&Ds and Clinical research organizations. More ...
One of her many illustrious achievements, is successful development of a Start-up R&D unit into reputable international research base, driving sales to over $ 5M within 3 years. She has also designed several award winning, quality compliant, cost- effective R&Ds and Clinical research organizations. More ...
Novel Technology Introductions
Shruti has successfully introduced over 35 different novel technologies within the Natural health products, Pharmaceuticals, OTC and Medical devices industry verticals. Some of the noted novel technologies she's facilitated include- Nano technology, Liposome entrapment, Mouth dissolve films, Medicated chewing gums, Needle free medicament administration etc. More ...
Shruti has successfully introduced over 35 different novel technologies within the Natural health products, Pharmaceuticals, OTC and Medical devices industry verticals. Some of the noted novel technologies she's facilitated include- Nano technology, Liposome entrapment, Mouth dissolve films, Medicated chewing gums, Needle free medicament administration etc. More ...
New Product Development
Shruti has successfully spearheaded drug product development programs for over 1500 formulations currently being sold in global markets including Canada, USA, Europe, BRIC, Africa and GCC (Gulf) countries. Shruti has worked with prescription pharmaceuticals, galenicals, vaccines, medical devices, modified release, biosimilars and platform delivery formulations. More ...
Shruti has led product development, testing and clinical studies of Natural health products (NHP), phyto-pharmaceuticals, health supplements, vitamins and minerals. More ...
Shruti has successfully spearheaded drug product development programs for over 1500 formulations currently being sold in global markets including Canada, USA, Europe, BRIC, Africa and GCC (Gulf) countries. Shruti has worked with prescription pharmaceuticals, galenicals, vaccines, medical devices, modified release, biosimilars and platform delivery formulations. More ...
Shruti has led product development, testing and clinical studies of Natural health products (NHP), phyto-pharmaceuticals, health supplements, vitamins and minerals. More ...
Audits And Quality Management
Shruti has conducted more than 500 Quality Audits and Due diligences globally. She has implemented Quality by design and Process analytical technique in R&D, production, packaging, quality departments, warehouses and logistics of a variety of industrial verticals including- pharmaceuticals, packaging, biotechnology, medical devices, foods, cosmetics, bulk drugs, chemicals and intermediates. More ...
Shruti has conducted more than 500 Quality Audits and Due diligences globally. She has implemented Quality by design and Process analytical technique in R&D, production, packaging, quality departments, warehouses and logistics of a variety of industrial verticals including- pharmaceuticals, packaging, biotechnology, medical devices, foods, cosmetics, bulk drugs, chemicals and intermediates. More ...
Continuous Improvement
Complementing her research and manufacturing expertise, she is also a noted Continuous Improvement expert. One of her most noted recent accomplishment is speedy turnaround of a struggling contract research and manufacturing company into a profitable business within nine months. She has implemented Continuous Improvement methodologies such as 5S, Lean, Kaizen, Lean six sigma, TQM, ISO, Just in time, Hoshin Kanri, Poka Yoke and more, for business processes within a variety of industrial verticals including- pharmaceuticals, packaging, biotechnology, medical devices, foods, cosmetics, bulk drugs, chemicals and intermediates. More ...
Complementing her research and manufacturing expertise, she is also a noted Continuous Improvement expert. One of her most noted recent accomplishment is speedy turnaround of a struggling contract research and manufacturing company into a profitable business within nine months. She has implemented Continuous Improvement methodologies such as 5S, Lean, Kaizen, Lean six sigma, TQM, ISO, Just in time, Hoshin Kanri, Poka Yoke and more, for business processes within a variety of industrial verticals including- pharmaceuticals, packaging, biotechnology, medical devices, foods, cosmetics, bulk drugs, chemicals and intermediates. More ...
Training
Shruti engages On-site and Webcast training workshops. She also speaks at Lunch and Learn training sessions for corporates and students. She provides powerful, elegantly simple framework with new tools and best-in-class industry practises for improving organizational performance and accelerating ROI.
Shruti engages On-site and Webcast training workshops. She also speaks at Lunch and Learn training sessions for corporates and students. She provides powerful, elegantly simple framework with new tools and best-in-class industry practises for improving organizational performance and accelerating ROI.
Below listed On-site and Webcast training workshops provide a powerful, elegantly simple framework with new tools and best-in-class industry practises for improving organizational performance and accelerating ROI.
The courses are available as standard modules as well as custom designed learning programs for each study group, executives and change leaders, to suit their requirements and business goals in the current competitive environment.
The courses are available as standard modules as well as custom designed learning programs for each study group, executives and change leaders, to suit their requirements and business goals in the current competitive environment.
Additional Honors & Awards
Pioneer – “Young Scientist Award” by PAMDAL – COLORCON for significant contribution to research in Pharmaceutical Sciences.
GOLD MEDAL and alumni achievers award from Prin. K.M.Kundnani College of Pharmacy Worli.
Searle India Performance Award
RESEARCH SCHOLAR – Ministry of Human Resources Development, Govt. Of India.
IDMA (Indian Drug Manufacturer’s Association) Research Award for the year 1991-92 for best research paper published in the faculty of Pharmaceutics.
IDMA (Indian Drug Manufacturer’s Association) Research Award for the year 1992-93 for best research paper published in the faculty of Pharmaceutics.
Late Smt. Jethibai H. Jagtiani Research Award and Certificate of Merit for – Sincerity, Quality and Quantity of Research work.
Late Smt. Kamla Balwani Award and Certificate of Merit for - Advanced Research work in Hi-Tech areas.
GOLD MEDAL and alumni achievers award from Prin. K.M.Kundnani College of Pharmacy Worli.
Searle India Performance Award
RESEARCH SCHOLAR – Ministry of Human Resources Development, Govt. Of India.
IDMA (Indian Drug Manufacturer’s Association) Research Award for the year 1991-92 for best research paper published in the faculty of Pharmaceutics.
IDMA (Indian Drug Manufacturer’s Association) Research Award for the year 1992-93 for best research paper published in the faculty of Pharmaceutics.
Late Smt. Jethibai H. Jagtiani Research Award and Certificate of Merit for – Sincerity, Quality and Quantity of Research work.
Late Smt. Kamla Balwani Award and Certificate of Merit for - Advanced Research work in Hi-Tech areas.
Publications
Controlled Release Isosorbide dinitrate pellets – Part 1 - Design and evaluation of controlled release capsule dosage form.
J. Pharmaceutical Sciences, 82 : 1288 ( 1993)
Controlled Release Isosorbide dinitrate pellets – Part 2 - In vivo studies.
J. Pharmaceutical Sciences, 82 : 1292( 1993)
HPTLC determination of etodolac in pharmaceutical formulations and human samples : HPTLC vs HPLC
A quantitative high performance thin layer chromatographic method for estimation of pseudoephedrine hydrochloride- application to stability studies
HPTLC determination of sulphamethoxazole and trimethoprim (Co-trimoxazole) from pharmaceutical formulations
Protracted disintegration time of hematinic capsules in soft gelatin shells : Part I : Gelatin – mineral interactions
Project for Glaxo (GSK) India.
Protracted disintegration time of hematinic capsules in soft gelatin shells : Part II : Gelatin – vitamin interactions
Project for Glaxo (GSK) India.
Awarded "Best Research paper" by IDMA.
Awarded "Best Research paper" by IDMA.
A look at process costing and cost-effectiveness of a pharmaceutical formulation.
Disintegration of soft gelatin haematinic capsules : role of pH, enzymes and other additives present in disintegration medium in reducing the disintegration time of stored samples.
Project for Glaxo India.
Microbial contamination of dyes and lake colors.
Project for M/s. Colorcon.
Non- pareil seeds - 2 3 factorial approach to optimization of coating for pellet preparation : coating pan Vs dish pelletiser.
Awarded "Best Research paper" by IDMA.
Non- pareil seeds – Application of Plackette Burman optimization design and concept of dissolution efficiency to pellet coating.
Mucosal delivery of peptide based therapeutics- Part 1 : Formulation considerations.
Mucosal delivery of peptide based therapeutics- Part 2 - Routes of administration
Modern developments in formulation technology.
Drugs from biotechnology.
Controlled Release formulations- Parts 1,2 and 3
SHRUTI'S PHILOSOPHY:
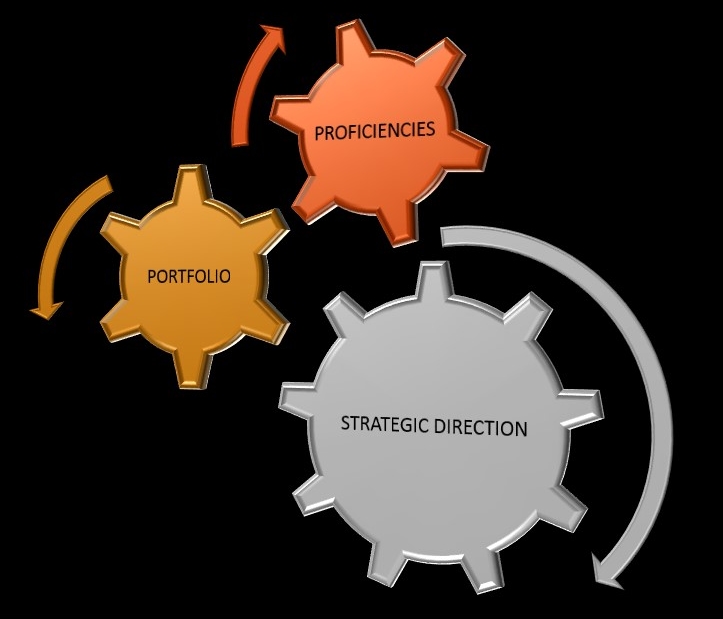
Only a coherent organization – one that pursues a clear strategic direction, builds a system of differentiating proficiencies, consistent with that direction and sells products/ services into demand markets that thrive within that system– can reliably and sustainably outpace competition.
Regardless of the industry, a company's power to win in every markets depends not just on external market positioning and not just on internal competencies, but on a COHERENT STRATEGY that aligns these at ENTRY LEVEL. Three interlocking elements make up a PROFITABLE BUSINESS: |
SHRUTI'S VALUES:
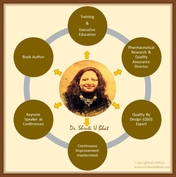 Roles of Dr. Shruti U Bhat
Roles of Dr. Shruti U Bhat 

COURSE MODULES:
Business Management topics:
Links
Continuous Improvement implementations by Dr. Shruti U ...
www.youtube.com/watch?v=4wGuXGyh--M
Sep 25, 2014 - Uploaded by Shruti Bhat
This video presents continuous improvement implementations byDr. Shruti U Bhat. Please review case ...Shruti U Bhat - YouTube
www.youtube.com/watch?v=UgyGA4Zk95s
Sep 25, 2014 - Uploaded by Shruti Bhat
Dr. Shruti U Bhat has implemented Quality by design for pharmaceutical product development through ...
|

VANCOUVER



VANCOUVER TIME
<!-Local Time Clock widget - HTML code - localtimes.info -->







 Time in Vancouver
Time in Vancouver 

Nice Blog and Good Info. Elmed Life Sciences manufacturer soft gelatin capsule where each soft gelatin capsule contains haematinic capsules
ReplyDelete