Org. Chem. Front., 2015, 2,1308-1312
DOI: 10.1039/C5QO00207A, Research Article
DOI: 10.1039/C5QO00207A, Research Article
D. Chandrasekhar, Satheesh Borra, Jeevak Sopanrao Kapure, Ghule Shailendra Shivaji, Gannoju Srinivasulu, Ram Awatar Maurya
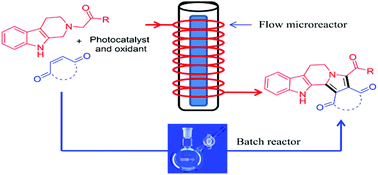
Fused [small beta]-carbolines were synthesized via a visible light photoredox catalyzed oxidation/[3 + 2] cycloaddition/oxidative aromatization reaction cascade in batch and flow microreactors.
Fused [small beta]-carbolines were synthesized via a visible light photoredox catalyzed oxidation/[3 + 2] cycloaddition/oxidative aromatization reaction cascade in batch and flow microreactors.
Fused β-carbolines were synthesized via a visible light photoredox catalyzed oxidation/[3 + 2] cycloaddition/oxidative aromatization reaction cascade in batch and flow microreactors.
Several structurally diverse heterocyclic scaffolds were obtained in good yields by coupling of tetrahydro-β-carbolines with a variety of dipolarophiles under photoredox multiple C–C bond forming events.
The photoredox coupling of tetrahydro-β-carboline with 1,4-benzoquinone was significantly faster in continuous flow microreactors and the desired products were obtained in higher yields compared to batch reactors.
Synthetic procedures General experimental procedures for the synthesis of N-alkylated of tetrahydro-β-carbolines 1a-f: In a 25 mL round bottom flask, tryptoline (86 mg, 0.5 mmol), α-halo carbonyls (0.5 mmol), Et3N (50 mg, 0.5 mmol) and CH2Cl2 (5 mL) was taken and the reaction mixture was stirred at ambient temperature for 2 h. Next the reaction mixture was diluted with CH2Cl2 (15 mL) and washed with water. The organic layer was dried over anhydrous Na2SO4 and evaporated to yield a crude product which was purified by silica-gel column chromatography using ethyl acetate/hexane in increasing polarity to yield compounds 1a-f.
General experimental procedures for the visible light photoredox catalyzed coupling of Nalkylated of tetrahydro-β-carbolines 1a-f with dipolarophiles 2a-g under batch conditions: In a 25 mL round bottom flask, tetrahydro-β-carbolines 1a-f (0.1 mmol), dipolarophiles 2a-g (0.1 mmol), [Ru(bpy)3Cl2]·6H2O (0.5 mol%) and MeCN (5 mL) was taken. The reaction vessel was kept at a distance of 10 cm (approx.) from a visible light source (11W white LED bulb) and the reaction mixture was stirred in open air condition until the reaction was complete (TLC). Next the reaction mixture was concentrated to give a crude product which was purified Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is © the Partner Organisations 2015 by silica-gel column chromatography using ethyl acetate/hexane in increasing polarity to yield compounds 3a-n
General experimental procedures for the visible light photoredox catalyzed coupling of Nalkylated of tetrahydro-β-carbolines 1a with dipolarophiles 2a in flow microreactors: A solution of tetrahydro-β-carboline 1a (0.2 mmol) and dipolarophile 2a (0.2 mmol) in MeCN (5 mL) was kept in one syringe and the solutions of photocatalyst [Ru(bpy)3Cl2]·6H2O (0.001 mmol in 5 mL MeCN) and t-BuOOH (2 mmol in 2 mL MeCN) were taken in two separate syringes. All the three solutions were pumped via two syringe pumps and mixed on an Xjunction and flown through the capillary microreactor wrapped over a visible light source (11W white LED bulb). Under stable conditions, exactly 6 mL of the reaction mixture was collected, concentrated to yield a crude product which was purified by silica-gel column chromatography using ethyl acetate/hexane in increasing polarity to yield compounds 3a
*Corresponding authors
aDivision of Medicinal Chemistry and Pharmacology, CSIR-Indian Institute of Chemical Technology, Hyderabad-500007, India
E-mail: ramaurya@iict.res.in
E-mail: ramaurya@iict.res.in
bNational Institute of Pharmaceutical Education and Research, Balanagar, Hyderabad-500035, India
cAcademy of Scientific and Innovative Research, New Delhi 110025, India
Org. Chem. Front., 2015,2, 1308-1312
DOI: 10.1039/C5QO00207A
///////






No comments:
Post a Comment